Adding a new control layer to CRISPR can "direct the site" to turn off oncogenes
2023-05-29 05:07:57
[China Pharmaceutical Network Technology News] At present, although scientists can use the CRISPR gene editing system to delete or replace any marker gene in living cells; but in the DNA repair process of cells, the damaged parts will be permanently deleted when they are glued together. A small number of genes cannot achieve precise control of gene editing.

As we all know, the CRISPR gene editing system is the current mainstream gene editing technology, scientists can use CRISPR to delete or replace any marker gene in living cells. It relies on a complex gene editing complex, including a DNA cleavage enzyme called "Cas9" and a "short RNA strand" that directs the enzyme to a specific region of the genome and directs Cas9 to cleave.
When Cas9 cleavage enzyme and RNA primers are introduced into the cell, specific cleavage can be performed in the genome; however, in the DNA repair process of the cell, a small part of the gene that is permanently deleted when the damaged part is bonded together cannot be Achieving precise control of gene editing has become an "imperfect flaw" in the technology.
Recently, MIT researchers have added an additional layer of control to the CRISPR gene editor through systematic response light, or precise control of gene editing. It is reported that with this new system, researchers only need to irradiate the target cells with ultraviolet light to achieve gene editing. This can help scientists study in more detail how cells and genetic material affect embryonic development and lead to genetic diseases; it can even provide a more targeted approach to accurately shut down oncogenes in tumor cells.
Sangeeta Bhatia, a professor at the Massachusetts Institute of Technology's HST Institute, the Koch Institute, and the Broad Institute, said the addition of any type of switch has the advantage of providing precise control over activation at time and space nodes.
The study was recently published in the German magazine Angewandte Chemie. The first author was Piyush Jain, a postdoctoral in medical science engineering at the Massachusetts Institute of Technology, and Professor Sangeeta Bhatia was the author of the article.
Light sensitivity
Before coming to the Massachusetts Institute of Technology, Dr. Piyush Jain developed a way to use light to control the process, called "RNA interference," in which small chains of RNA are delivered to cells to temporarily block Specific genes. When he saw the emergence of CRISPR technology at MIT, he was thrilled and hoped to use light to activate the CRISPR system in the same way.
Professor Bhatia said that the researchers have made many efforts to create a photosensitive CRISPR system, including changing some of the properties of the Cas9 enzyme to start shearing only at certain wavelengths of light; but the MIT team decided A different approach is taken to transform the RNA primers for light sensitivity.
Professor Bhatia added that in addition to adding a photo-activated protector, you don't need anything else. This attempt makes the system more modular. Since the future of the CRISPR system has broad application prospects in humans, MIT's modification of the RNA primer chain is obviously much easier than the production of photosensitive Cas9 in target cells.
In order to make the guide RNA photosensitive, the MIT team created a "protector" - a DNA sequence that cleaves the bond to form the backbone. These DNA segments can be directed to bind to different guide RNAs to form a complex, thereby preventing RNA primers from being linked to other target genes.
When the researchers exposed the target cells to light at a wavelength of 365 nm (in the ultraviolet range), the DNA "protector" cleaves into several smaller fragment fragments and sheds from the RNA, allowing the RNA to bind to the marker gene, guiding It was cleaved by Cas9 endonuclease.
Targeting multiple genes
In this study, the researchers confirmed that light is used to control the editing of green fluorescent protein (GFP) genes, two of which are often found on the cell surface of some overexpressing cancers.
Professor Bhatia said that if this is a law-abiding project, scientists will design protection sequences for different target sequences. Their team has designed protectors for different genes and found that they can be activated in this "light control" way. In addition, in a multiplexed experiment, when a hybrid type of protector is used, only the target is cleaved in the case of exposure.
In the view of Professor Bhatia, this can precisely control the time nodes of gene editing, help researchers study the timing of cell activities (including disease progression, etc.), and bring hope to determine the optimal timing of intervention by shutting down genes, Bhatia The laboratory is also exploring the clinical application of this technology.
James Dahlman, assistant professor of biomedical engineering at Georgia Tech, did not participate in the study. He said that CRISPR-Cas9 is a powerful technology that scientists can use to study how genes affect cellular activity. This important advancement will precisely control genetic changes. Therefore, this research provides the scientific community with a very useful tool to improve the optimization of many genetic editing.
It is reported that it is highly probable that it will be used to close the oncogene in skin cancer because the skin is easily exposed to ultraviolet light. The Bhatia team is working on a "general protector" to apply to any RNA-guided segment, avoiding the need to design a protector for each RNA, and inhibiting CRISPR-Cas9 from simultaneously cleavage of multiple targets. Analysts believe that through the efforts of scientists, this technology will eventually go to clinical medicine, improve people's lives, and hope to cure skin cancer.
Currently, the research has been funded by the Ludwig Center for Molecular Oncology, the Marie-D and Pierre Casimir-Lambert funds, the Koch Institute fund from NCI, and the Marble Center for Cancer Nanomedicine.
Editor added:
The combination of optogenetics and genome technology, which requires the use of engineered nucleases, provides us with an opportunity to manipulate any gene with light.
The application and popularization of gene editing technology in human gene therapy has become an irresistible trend. With more in-depth research and development, its commercial value cannot be underestimated. In addition, precision medicine has been selected into the "Thirteenth Five-Year Plan" project this year, and has risen to a national strategy, which has brought a strong catalytic effect to the development of China's genetic editing technology.

As we all know, the CRISPR gene editing system is the current mainstream gene editing technology, scientists can use CRISPR to delete or replace any marker gene in living cells. It relies on a complex gene editing complex, including a DNA cleavage enzyme called "Cas9" and a "short RNA strand" that directs the enzyme to a specific region of the genome and directs Cas9 to cleave.
When Cas9 cleavage enzyme and RNA primers are introduced into the cell, specific cleavage can be performed in the genome; however, in the DNA repair process of the cell, a small part of the gene that is permanently deleted when the damaged part is bonded together cannot be Achieving precise control of gene editing has become an "imperfect flaw" in the technology.
Recently, MIT researchers have added an additional layer of control to the CRISPR gene editor through systematic response light, or precise control of gene editing. It is reported that with this new system, researchers only need to irradiate the target cells with ultraviolet light to achieve gene editing. This can help scientists study in more detail how cells and genetic material affect embryonic development and lead to genetic diseases; it can even provide a more targeted approach to accurately shut down oncogenes in tumor cells.
Sangeeta Bhatia, a professor at the Massachusetts Institute of Technology's HST Institute, the Koch Institute, and the Broad Institute, said the addition of any type of switch has the advantage of providing precise control over activation at time and space nodes.
The study was recently published in the German magazine Angewandte Chemie. The first author was Piyush Jain, a postdoctoral in medical science engineering at the Massachusetts Institute of Technology, and Professor Sangeeta Bhatia was the author of the article.
Light sensitivity
Before coming to the Massachusetts Institute of Technology, Dr. Piyush Jain developed a way to use light to control the process, called "RNA interference," in which small chains of RNA are delivered to cells to temporarily block Specific genes. When he saw the emergence of CRISPR technology at MIT, he was thrilled and hoped to use light to activate the CRISPR system in the same way.
Professor Bhatia said that the researchers have made many efforts to create a photosensitive CRISPR system, including changing some of the properties of the Cas9 enzyme to start shearing only at certain wavelengths of light; but the MIT team decided A different approach is taken to transform the RNA primers for light sensitivity.
Professor Bhatia added that in addition to adding a photo-activated protector, you don't need anything else. This attempt makes the system more modular. Since the future of the CRISPR system has broad application prospects in humans, MIT's modification of the RNA primer chain is obviously much easier than the production of photosensitive Cas9 in target cells.
In order to make the guide RNA photosensitive, the MIT team created a "protector" - a DNA sequence that cleaves the bond to form the backbone. These DNA segments can be directed to bind to different guide RNAs to form a complex, thereby preventing RNA primers from being linked to other target genes.
When the researchers exposed the target cells to light at a wavelength of 365 nm (in the ultraviolet range), the DNA "protector" cleaves into several smaller fragment fragments and sheds from the RNA, allowing the RNA to bind to the marker gene, guiding It was cleaved by Cas9 endonuclease.
Targeting multiple genes
In this study, the researchers confirmed that light is used to control the editing of green fluorescent protein (GFP) genes, two of which are often found on the cell surface of some overexpressing cancers.
Professor Bhatia said that if this is a law-abiding project, scientists will design protection sequences for different target sequences. Their team has designed protectors for different genes and found that they can be activated in this "light control" way. In addition, in a multiplexed experiment, when a hybrid type of protector is used, only the target is cleaved in the case of exposure.
In the view of Professor Bhatia, this can precisely control the time nodes of gene editing, help researchers study the timing of cell activities (including disease progression, etc.), and bring hope to determine the optimal timing of intervention by shutting down genes, Bhatia The laboratory is also exploring the clinical application of this technology.
James Dahlman, assistant professor of biomedical engineering at Georgia Tech, did not participate in the study. He said that CRISPR-Cas9 is a powerful technology that scientists can use to study how genes affect cellular activity. This important advancement will precisely control genetic changes. Therefore, this research provides the scientific community with a very useful tool to improve the optimization of many genetic editing.
It is reported that it is highly probable that it will be used to close the oncogene in skin cancer because the skin is easily exposed to ultraviolet light. The Bhatia team is working on a "general protector" to apply to any RNA-guided segment, avoiding the need to design a protector for each RNA, and inhibiting CRISPR-Cas9 from simultaneously cleavage of multiple targets. Analysts believe that through the efforts of scientists, this technology will eventually go to clinical medicine, improve people's lives, and hope to cure skin cancer.
Currently, the research has been funded by the Ludwig Center for Molecular Oncology, the Marie-D and Pierre Casimir-Lambert funds, the Koch Institute fund from NCI, and the Marble Center for Cancer Nanomedicine.
Editor added:
The combination of optogenetics and genome technology, which requires the use of engineered nucleases, provides us with an opportunity to manipulate any gene with light.
The application and popularization of gene editing technology in human gene therapy has become an irresistible trend. With more in-depth research and development, its commercial value cannot be underestimated. In addition, precision medicine has been selected into the "Thirteenth Five-Year Plan" project this year, and has risen to a national strategy, which has brought a strong catalytic effect to the development of China's genetic editing technology.
Boob tape, breast lift tape
Boob tape is based on 95% cotton and 5% Elastic Spandex fabric. The material is safe, soft, breathable, which makes you comfortable in it. It has super strong elasticity, lateral stretching is more than 230%. It is coated with imported wave pattern glue, which ensures the tape has stable adhesion and easy to remove, do not hurt your skin. It has a certain waterproof effect, the adhesion will not be reduced even you are sweaty or swimming. It has wonderful lifting and gathering effect, let you have a proud chest curve.

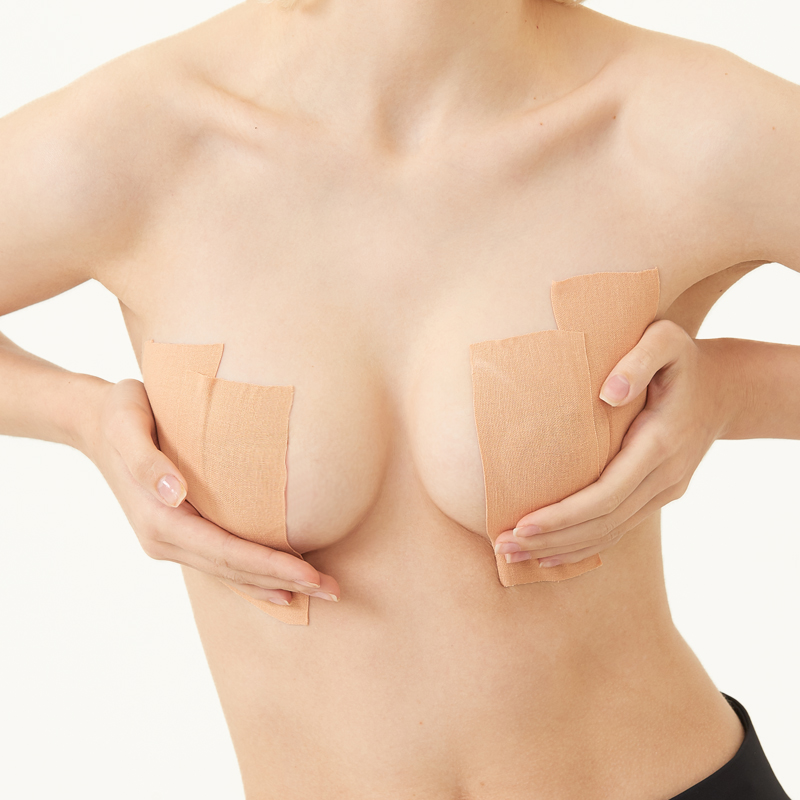
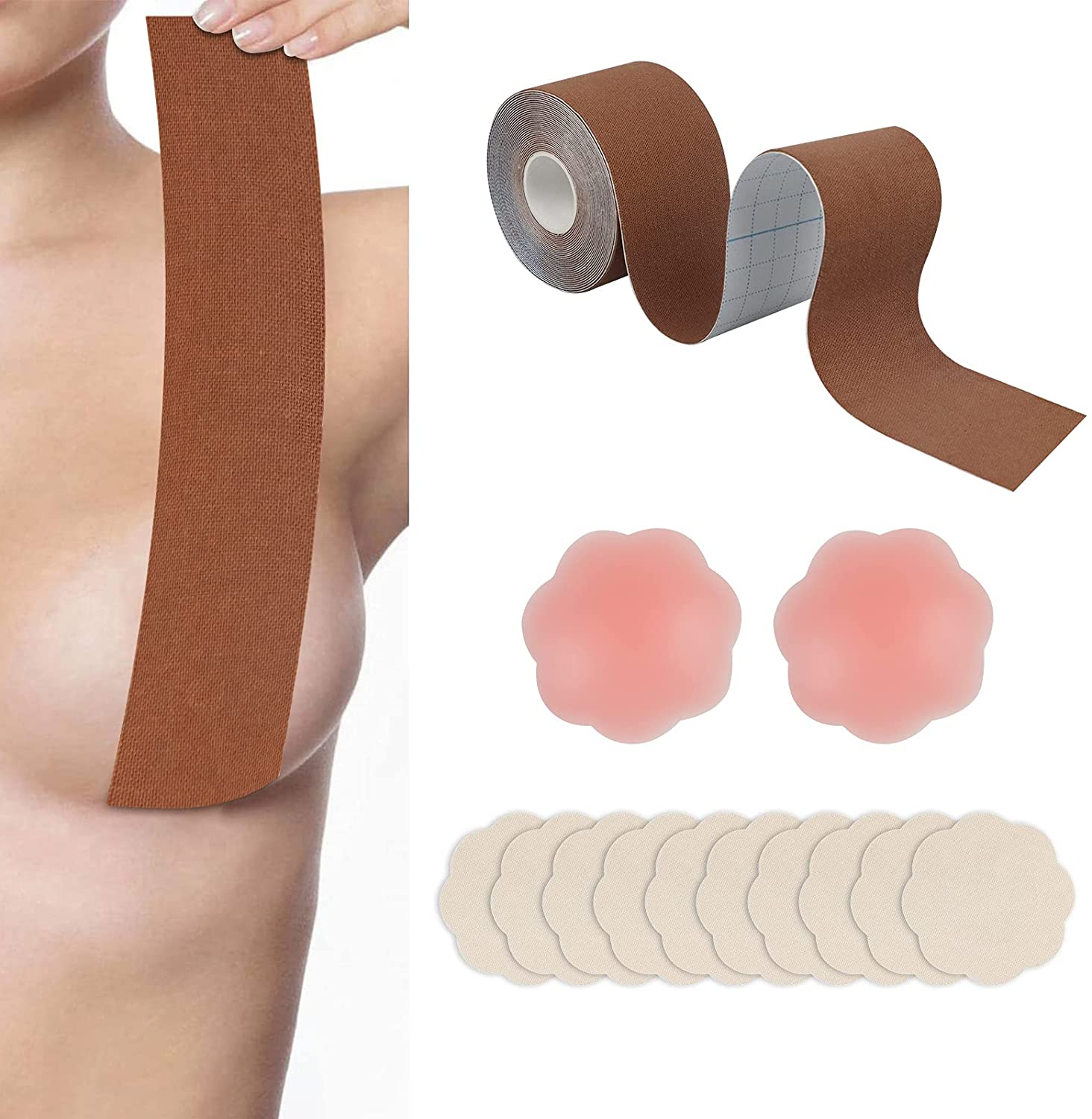
boob tape,breast lift tape,boob tape and nipple cover,boob lift tape,breast tape
Kunshan Jieyudeng Intelligent Technology Co., Ltd. , https://www.jerrytape.com