Talking about the Strategic Investment Prospect of Gene Therapy Drug Market
Wei Si Teng Diagnosing Zhou Yong: Talking about the Strategic Investment Prospect of Gene Therapeutic Drug Market
(This article is the original founder of Wei Si Teng diagnosis, Zhou Yong original, reproduced need to indicate the source)
Gene therapy (genetherapy): refers to (normal or wild-type) gene into human cells to exert a biological effect, so as to achieve the purpose art methods of treating diseases.
Gene therapy is one of the most revolutionary medical technologies developed with the maturity of DNA recombination technology and gene cloning technology in the 1970s and 1980s. It is a biomedical treatment based on the alteration of human genetic material. It shows a unique advantage in the treatment of major diseases. After more than 30 years of development, gene therapy has been expanded from the initial treatment for single-gene hereditary diseases to more than 6,000 diseases such as AIDS, hepatitis B, cancer, sickle anemia, hemophilia, mucopolysaccharidosis III Type, hereditary psychosis, infectious diseases, cardiovascular diseases, autoimmune diseases and metabolic diseases. The breakthrough in gene therapy technology has made gene therapy one of the top 10 breakthroughs in Sicence 2009 and published a column called "A Comeback for Gene Therapy."
In general, therapeutic genes cannot be directly introduced into human cells, and this process requires the use of a certain carrier (tool). Commonly used vectors are classified into viral vectors and non-viral vectors. In the current study, the most commonly used vector (tool) is a viral vector. All viruses have the ability to attack host cells and introduce genetic material into their host cells. The genetic material of the virus, the genome of the virus, is able to "kidnap" the normal protein expression system of the host cell to produce viral proteins preferentially, thereby enabling the virus to self-replicate rapidly.
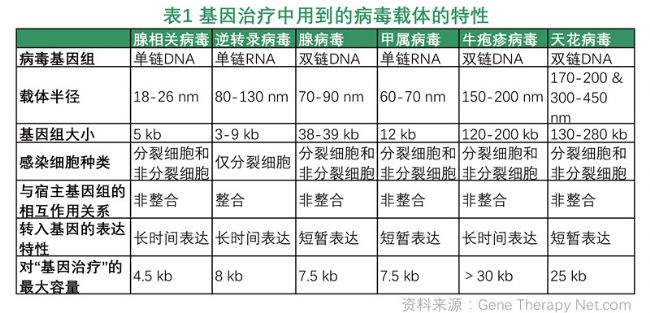
With the development of technology, adeno-associated viruses and retroviruses are mainly used in clinical research of gene therapy. The adeno-associated virus has been shown to be a virus that is completely harmless to the human body and that causes an immune response that is not apparent. The virus can treat "therapeutic genes" as much as in host cells for long-term expression. This vector is more suitable for the treatment of gene defects in cells that do not divide for a long time, such as ophthalmic diseases or cardiovascular diseases caused by genetic variation. . Retroviruses are able to integrate "therapeutic genes" into the host genome, enabling "good genes" to be delivered to the next generation of cells in which the host cells divide or differentiate. Therefore, retroviral vectors are often used for genetic modification of hematopoietic stem cells, often for the treatment of blood-related genetic diseases or cancer.
1 gene therapy administration and target cells
Current clinical studies of gene therapy are directed to the genetic modification of somatic cells. From the perspective of gene introduction, it can be divided into two ways: in vitro introduction and in vivo introduction:
In vitro introduction : refers to the cells that need to be genetically modified (such as hematopoietic stem cells, blood cells, etc.) are extracted from the patient, mixed with the viral vector in vitro, and the genetic modification of the extracted cells is completed. The modified cells are then reinfused into the patient's body.
In vivo introduction : refers to the way of injecting the carrier of the disease into the lesion of the patient directly by injection or the like.
From the introduced target cells, it is divided into two types, somatic cells and germ cells. The effect of gene therapy for somatic cells is limited to the patient himself, and the altered genes are not inherited to the offspring, and have certain controllability in genetics and ethical risks. Therefore, somatic cell introduction is the mainstream clinical research method. In the United States, there are now more than 600 gene therapy clinical trials for somatic cell introduction in progress. Most clinical trials target diseases caused by single-gene mutations such as AIDS, hemophilia, thalassemia, and cystic fibrosis. The genetic modification of germ cells can theoretically solve the genetic defects of patients more efficiently, and make normal genes can be passed on to future generations, but in view of ethical issues and maturity of technology, in many countries, such as Australia, Canada, Germany, Israel, Switzerland, the Netherlands and other legislation have strictly prohibited gene therapy for germ cells. Although there is no relevant legislation in the United States, the current research is mainly focused on the field of gene therapy for somatic cells.
2 Gene therapy for cancer
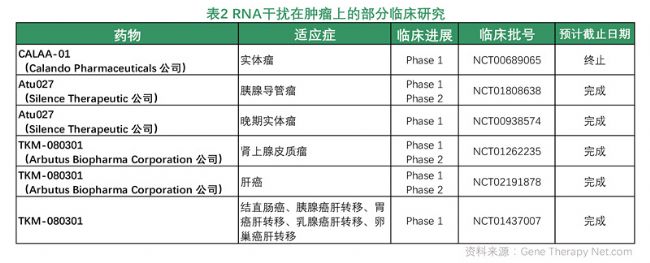
Gene silencing therapy: RNA interference (RNAi) refers to the highly conserved, highly-specific degradation of homologous mRNA induced by double-stranded RNA (dsRNA) during evolution. Studies have shown that some important signaling pathways and genes play a key role in the development of tumors. Therefore, the application of RNAi technology to reduce the expression levels of key molecules and some important oncogenes in these signaling pathways is a very effective strategy. RNAi can specifically inhibit oncogene expression in cancer cells without affecting gene expression in normal cells. Among them, small interfering RNA (siRNA) can produce multi-gene silencing effects. From the discovery to the present, many scholars, biotech companies and pharmaceutical companies have begun to widely use siRNA to treat tumors. In 2008, Calando Pharmaceuticals of the United States applied for the siRNA drug CALAA-01 for solid tumors, which was approved by the FDA to enter the clinical phase I trial. It is the first clinical trial of siRNA drugs for cancer treatment. In July 2009, the first phase clinical trial of RNA interference targeting solid tumors conducted by Silence Therapeutics of Germany showed that after 24 patients were treated with Atu027 multiple times, 9 patients were stable and 6 had good results. The remaining patients are receiving treatment.
Tumor suppressor gene therapy: tumor suppressor genes, also known as anti-cancer genes, have the effect of inhibiting cell proliferation when activated, but can inhibit or eliminate tumor suppression after being inhibited or lost under certain circumstances. effect. More than 20 tumor suppressor genes, such as the p53 gene, have been isolated and cloned.
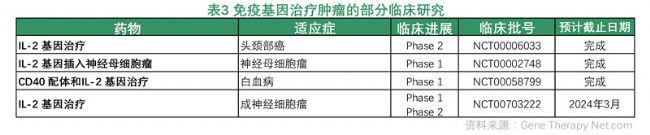
Immune gene therapy for tumors: The most commonly used tumor immunogene therapy is the introduction of cytokines into somatic cells to create a microenvironment that enhances the anti-tumor immune response, thereby achieving tumor treatment.
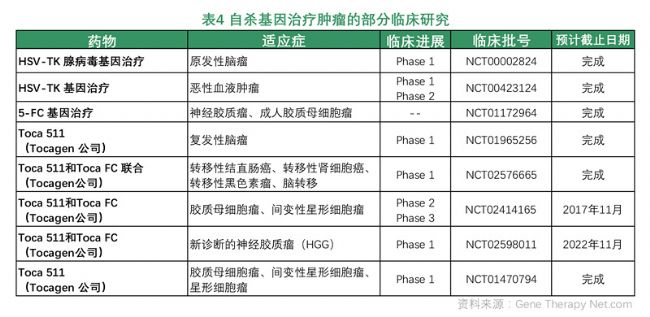
Suicide gene therapy: Suicide gene refers to the introduction of certain viruses or bacterial genes into target cells, and the expressed enzymes can catalyze the conversion of non-toxic prodrugs into cytotoxic substances, thereby causing the gene to be carried. The receptor cells are killed and such genes are called suicide genes. There are many types of suicide gene therapy systems, including herpes simplex virus thymidine kinase/glycoguanine (HSV-tk/GCV) system, cytosine deaminase/5-fluorocytosine (CD/5-FC) system, Herpes zoster virus thymidine kinase/arabose methoxypurine (VZV2tk/Ara2M) system, nitroreductase/CB1954 (NTR/CB1954) system, ICE gene, and the like.
Tocagen's Toca 511 and Toca FC combination therapy is licensed by the FDA orphan drug for the treatment of malignant glioma. In this combination therapy, Toca 511 is a retroviral replication vector that selectively delivers the gene for cytosine deaminase (CD) into cancer cells. After Toca511 is spread over the tumor, cancer cells that express the CD gene can convert fluorocytosine (5-FC) into the anticancer drug 5-fluorouracil (5-FU), which in turn mediates tumor cell programmed cell death and enhances the body. An immune response to a tumor. The Toca 511 and Toca FC combination therapy was conducted in Phase II and Phase III clinical trials in 2015. According to the clinical interim report, the median survival rate of patients with malignant glioma after the combination therapy reached 13.8 months, which was better than the mid-term of 7 months. The historical benchmark of survival rate showed good clinical efficacy.
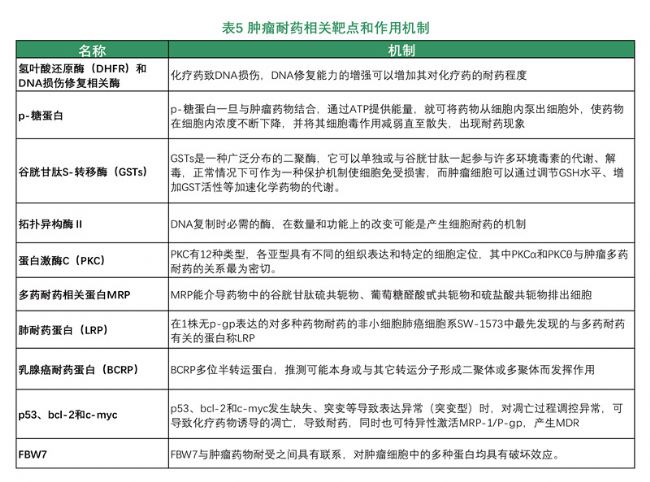
Tumor resistance gene therapy: Chemotherapy is one of the most important methods for the treatment of malignant tumors in the clinic. However, because tumor cells often become resistant to chemotherapy drugs, patients are no longer sensitive to treatment, which ultimately leads to chemotherapy failure and even disease recurrence. According to the drug resistance characteristics of tumor cells, drug resistance can be divided into two categories: drug resistance (PDR) and multidrug resistance (MDR). Prodrug resistance (PDR) refers to the sensitivity of different anti-tumor drugs to non-identical drugs; multi-drug resistance (MDR) refers to some cancer cells against an anti-tumor The drug produces drug resistance, and it also develops resistance to other non-similar drugs, which is the main reason for the failure of tumor chemotherapy (chemotherapy).
Tumor treatment of oncolytic viruses: It has been found that the use of viruses can treat tumors, and many viruses have been found to have natural oncolytic properties, such as influenza virus, herpes simplex virus, West Nile encephalitis virus, Newcastle disease virus, and vaccinia virus. Since the pathogenicity of wild-type and attenuated strain viruses is difficult to control artificially, these viruses cannot be applied to the clinical treatment of cancer. With the development of virology and genetics, the functions and mechanisms of various viral genes have become increasingly clear, and with the advancement of technologies such as genetic engineering, people have been able to perform various directional operations and transformations on viral genes, thereby changing directionally. And control the behavior and function of the virus. In 1991, herpes simplex virus type 1 (HSV-1) was genetically engineered to inactivate the Thymidine kinase (TK) gene, which inhibited cancer cells and autonomously replicated. Oncolytic virus strain. Later, strategies and research to treat cancer using oncolytic viruses have developed rapidly. Following HSV-1, recombinant gene viruses such as adenovirus and vaccinia virus have been further developed; in addition, studies on genetic modification of wild-type viruses or naturally-mutated attenuated strains have also achieved good results.
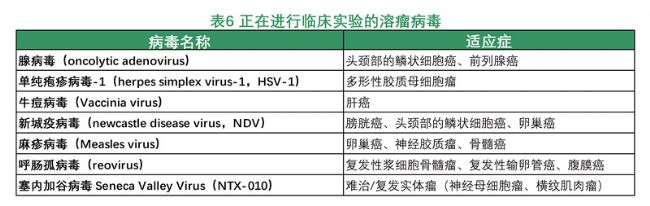
Compared with other projects that are still in clinical phase I and phase II, the field of cancer will become a therapeutic field that is expected to list more products in the near future. Breast cancer, cervical cancer, rectal cancer, lung cancer, melanoma, neuroblastoma, ovarian cancer, pancreatic cancer, prostate cancer, kidney cell cancer and many other tumor fields are the most promising products. In particular, melanoma, head and neck cancer and prostate cancer are considered to be the most promising in the United States.
According to IMS data, the global anti-cancer drug market will be 100 billion US dollars in 2014 and will increase to 150 billion US dollars in 2020. Anti-tumor monoclonal antibody drugs with targeted, specific effects and low toxicity accounted for 34%, reaching $23.8 billion. The price of a monoclonal antibody is similar to the price of a gene therapy. Among them, Herceptin, a famous monoclonal antibody for breast cancer, is a heavy product for the treatment of breast cancer. In 2013, its global sales reached 6.839 billion US dollars, and its sales in 2014 reached 6.27 billion US dollars. If gene therapy is effective on cancer, it can replace the monoclonal antibody, and its market is expected to be $23.8 billion.
3 Third generation gene editing technology: CRISPR-Cas9
Clustered regular interspaced short palindromic repeats/CRISPR associated proteins: The third generation of gene editing technology enables simultaneous editing of multiple genes with higher targeting efficiency. The operation is simple, the construction cost is low, and the editing efficiency is high. At present, CRISPR is more than just a multi-purpose scissors that can cut any gene precisely, and it can be used as a multifunctional transmission system to precisely control any gene.
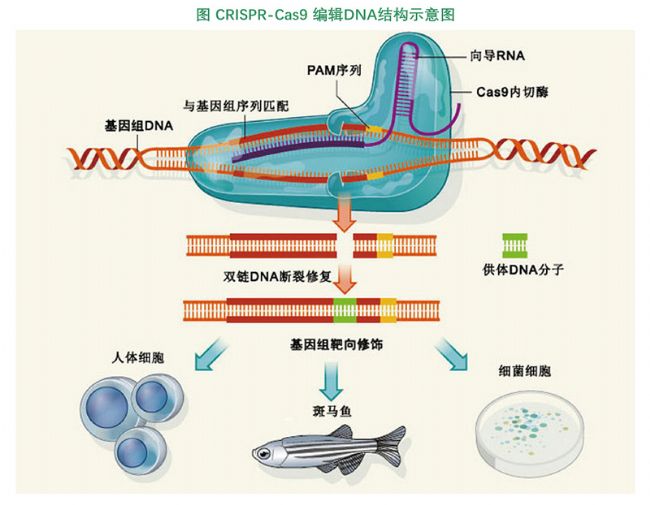
The widely used CRISPR/Cas9 system can be mainly divided into two different applications in vitro and in vivo. For in vitro applications, purified Cas9 nucleases are coupled with guide RNA for targeted cleavage of genomic fragments in vitro to replace conventional restriction enzymes or PCR clones. There are two main combinations for in vivo applications, one is the active form, the purified Cas9 nuclease with nuclear localization signal, and the purified guide RNA, which enters the nucleus and exerts directional shear on the genome; The Cas9 and the guide RNA are delivered to the cells in the form of a vector, which is expressed and expressed in vivo. Two in vivo applications are gene editing that enables gene knockout and insertion primarily at the cellular or individual level.
4 Combination of CRISPR and Cell Therapy (CAR-T): The most advanced oncology technology
Chimeric antigen receptor (CAR) technology: an artificially designed antibody that targets a tumor-associated antigen or a tumor-specific antigen single-chain antibody-CAR by genetic engineering into T cell surface and redirects T cells. The reaction allows T cells to recognize tumor cells in a non-HLA molecularly restricted manner and exert a killing effect. Problems with CAR-T: CAR-T has the advantages of high specificity, significant curative effect, and low side effects. Its common side effects include cytokine release syndrome, off-target toxicity, etc. Difficulties in application include poor treatment of solid tumors and inability to supply Spot.
At present, with the help of CRISPR technology, CAR-T technology has been successfully upgraded: CRISPR technology can efficiently edit multi-point T cells, effectively solving the problems of strong side effects, poor solid tumors, large differences in individual efficacy, and difficulty in commercialization. The combination of CRISPR and CAR-T is expected to rapidly advance clinical trials and achieve industrial production.
Foreign CAR-T therapy giants have invested in CRISPR technology: Almost all CAR-T therapy companies are currently cooperating with gene editing companies, including six CAR-T therapy companies including Novartis, Juno and Cellectis, and Intellia Therapeutics and Editas Medicine. Developed a combination of CRISPR technology and CAR-T therapy in collaboration with gene editing companies such as CRISPR Therapeutics. Novartis is the fastest growing company in the world for CAR-T technology development. It is expected that Novartis's CTL019 first indication for acute lymphoblastic leukemia will be approved in 2016, and will receive approval for chronic lymphocytic leukemia and non-Hodgkin in 2017. Two indications such as lymphoma. Novartis and CRISPR technology giant Intellia launched a five-year R&D cooperation program to better integrate CRISPR and CAR-T, and accelerate the development of CRISPR/Cas9 technology in the application of CAR-T cell therapy in hematopoietic stem cells. .
In the course of gene therapy, especially in malignant tumors, only the vector can be injected directly into the tumor. If injected intravenously, the carrier will be cleared quickly and it will be difficult to achieve therapeutic effects. Scientists have designed different protocols, adapted the structure of the vector, and constructed different types and characteristics of targeting vectors. The target transduction vector is constructed by cross-linking different ligand components onto the outer membrane of the viral vector; or introducing a cis-regulatory element into the DNA of the vector, constructing a targeted transcription vector, and the like. The construction of these vectors strongly promotes the development of gene therapy.
Insertion of the viral vector or randomization into the genome has the potential to cause insertional mutagenesis and activation of oncogenes. For example, the AAV2 adeno-associated virus, which was thought to be harmless in the past, is actually associated with hepatocellular carcinoma. Although AAV2 DNA insertion is less likely to cause cancer, it should also be highly valued. In 2000, patients at the University of Pennsylvania were treated with gene therapy. In the death of the disease; in 2002, in France, congenital immunodeficiency bubble boy gene therapy clinical trials of leukemia side effects. Both failed cases were attributed to the insecurity of the viral vector.
The viral vector is highly immunogenic and has significant cytotoxicity at high titers. The solution is to use non-viral delivery systems, such as cationic polymer carriers, nanoparticle carriers, liposomes, polyethyleneimine and other biocompatible carriers will become a promising gene delivery system. Non-viral delivery systems have shown advantages in the application of gene therapy for genetic diseases, and are increasingly used to treat malignant tumors, infectious diseases, and tissue engineering research.
Ideal gene therapy should be able to regulate the proper expression of the therapeutic gene depending on the nature and severity of the disease. However, compared with the gene therapy vector system, the study of the regulation of gene expression regulation is relatively lagging. A method such as linkage gene amplification can be used to appropriately increase the copy number of the foreign gene in the cell, and the control signal for gene expression such as a promoter or an enhancer. I believe that with the development of human genomics, the problem will be solved very well.
5 The future of gene therapy
Gene therapy is widely used With the development of gene therapy technology, gene therapy has been expanded from the initial treatment for single-gene hereditary diseases to more than 6,000 diseases such as AIDS, hepatitis B, cancer, sickle anemia, hemophilia, mucopolysaccharide Storage disease type III, hereditary psychosis, infectious diseases, cardiovascular diseases, autoimmune diseases and metabolic diseases. At present, gene therapy is undergoing diseases ranging from rare diseases to non-rare diseases, and eventually to the development of common diseases. The pathogenesis of the disease is also from simple to complex. With the advancement of treatment for common diseases, gene therapy technology will become increasingly mature and achieve huge profits.
The gene therapy technology led by CRISPR is booming. In December 2015, the US Science published its top 10 scientific breakthroughs in 2015. The CRISPR genome editing technology was selected as the number one breakthrough this year. Gene therapy is favored by countries At the end of 2015, the White House once again released the "New Strategy for American Innovation". The report clearly defines precision medicine including gene therapy as the future development strategy of the United States in the medical field. China's precision medical plan will be launched in 2016, with a planned investment of 60 billion by 2030. Under the background of optimistic precision medicine in the world, the market is full of confidence in gene therapy related companies. Gene therapy is concerned by the capital market for gene therapy-related pharmaceutical companies, with fewer listed products (even without a product), and large investment in research, all affect the company's profitability. But it does not affect the enthusiasm of the capital market. The gene therapy company's performance on the stock market is eye-catching: the gene therapy leader Blubirdbio has been on the market for a year and a half, and its share price has risen tenfold. Editas Medicine, founded by the inventor of the company, Zhang Feng, rose more than 130% in the first month of listing. In addition, gene therapy-related companies are actively starting IPOs. On May 16, 2016, Editas Medicine and the Cystic Fibrosis Foundation Therapeutics (CFFT) announced a three-year, $5 million partnership; on the same day, Caribou also Announced good news, the company completed $30 million Series B round of financing, new investors including Anterra Capital, Heritage Group, Maverick Capital Ventures and Pontifax AgTech, the original investors involved in this round of financing include F-Prime Capital Partners, Novartis, Mission Bay Capital and 5 Prime Ventures.
Major world-class pharmaceutical companies have accelerated the deployment of world-class international pharmaceutical companies into the field of gene therapy in various ways, such as cooperation, purchasing, investment and independent research and development. Among them, the successful listing of GSK gene product Strimvelis also set a successful example for the development of gene therapy: the project was funded by the non-profit organization (Telethon Foundation of Italy) and completed by the world-class pharmaceutical company GSK. , from research and development to listing. In 2011, pharmaceutical giant Amgen acquired the Imlygic developed by Biovex for the price of $425 million and the $575 million milestone. In October 2015, the FDA officially approved the listing.
Probable market catalysts in the future A large number of clinical phase III experimental data will be disclosed: As of the end of 2013, about 5% of the nearly 2,000 gene therapy trials reached the phase III clinical phase. After three years of experimental cycles, the batch three phase trials are about to be completed. . The investment in gene therapy by world-class pharmaceutical companies will speed up the process of product launch: cooperation with world-class pharmaceutical companies and biotech companies and research institutes led by GSK will take over from the clinical phase III to complete commercial transformation. Spark Therapeutics' GM treatment program for RPE65 hereditary retinal dystrophy was approved by the US FDA in 2016. Novartis CAR-T therapy, Juno Therapeutics' CAR-T treatment product JCAR15 for adult acute lymphoblastic leukemia and Kite's KTE-C19 for diffuse large B-cell lymphoma are expected to be submitted for marketing in 2016. It is worth noting that A combination of CAR-T and CRISPR technology.
(This article is the original founder of Wei Si Teng diagnosis, Zhou Yong original, welcome to reprint, please indicate the source)
[ Author : Zhou Yong, founder/Chairman of Gaosheng Pharmaceutical, founder/Chairman of Weisseng Diagnostics, co-founder of Gaochuanghui Biomedical Transformation Platform, Vice President of Beijing Haidian District Biological and Health Industry Association, Chongqing Vice President of the Municipal Pharmaceutical Industry Association, Chongqing High-level Talents Special Support Program, and Chongqing Innovation and Entrepreneur Leading Talents]
Increasing complexity of operation theater/ICU equipment requires medical pendant more expansibility, reliable, convenience. Lewin series medical pendant combined LEWIN abundant operation room/ICU equipment produce and integrate experience, the design follows human body enginerring principle; The ceiling pendnat function fits doctor and patient's actual using demands, pay attention to convenient actual operation, can meet different environment demands, has highly cost-effective;

Medical Pendant,Surgical Pendant,Hospital Pendant,Cavascope Pendant Anesthesia Pendant,Ceiling Pendant,Hospital Pendant,Medical Gas Pendant
Shandong Lewin Medical Equipment Co., Ltd. , https://www.lewinmed.com